Secret Recipes Continued
Part 2 - Details
The rest of my 2006 presentation ventured into some controversial territory concerning the psychoactivity of ergine and isoergine. The problem, which remains still, is that Hofmann and a few others who had tested pure ergine and/or isoergine on themselves usually reported very little or at best mild psychedelic effects.
I wanted to devise a hypothesis that would cover both the hydrolysate of ergot for Eleusis, and the ololiuqui of Central American shaminic use. As for the latter, Jonathan Ott wrote to us for the “Mixing the kykeon“ paper:
Ololiuqui was far more prominent as an entheogen here in Mesoamerica than those mushrooms—the mushrooms are mentioned only here and there by a few competent chroniclers; yet almost an entire book was devoted to denouncing mainly the ololiuhqui idolatry. The annals of the Inquisition contain many times more autos de fe for ololiuqui than for mushrooms.
If ergine or isoergine alone seemed not up to the task of providing a psychedelic experience for some of us moderns, a logical hypothesis to propose would be that it was the method of preparation at Eleusis or for the American shamans that did the trick. Inevitably, during such preparation processes, an equilibrium mixture of ergine and isoergine would be produced. I decided therefore to suggest in my presentation that this equilibrium mixture was more productive of psychedelic experience than either alkaloid alone. As I saw no immediate reason why that should be so, I explored the chemical literature and found an article in Tetrahedron1 that described some fascinating properties of the ergoline ring system. I presented several drawings, some animated, showing how the ergolene ring system could take on different conformations and suggested why the more stable ones might explain an increase in psychoactivity, particularly for isoergine.
My presentation was designed to appeal to the type of audience I thought would be present: youngsters of all ages, psychedelic enthusiasts all, a few well versed in chemistry but most not so, but perhaps receptive of a visually stimulating look at these fascinating molecules. It was suggested by an expert or two, however, that I had gone a bit too far in my interpretations of the known facts and mechanisms! I would plead, in retrospect, that the Tetrahedron article most certainly supplies some clues worth following up, but that I used them somewhat unreservedly, more for entertainment value than as something I might publish in the scientific literature.
So then, on with the original presentation, after which I shall provide some commentary and updates that have come to mind in the intervening years.
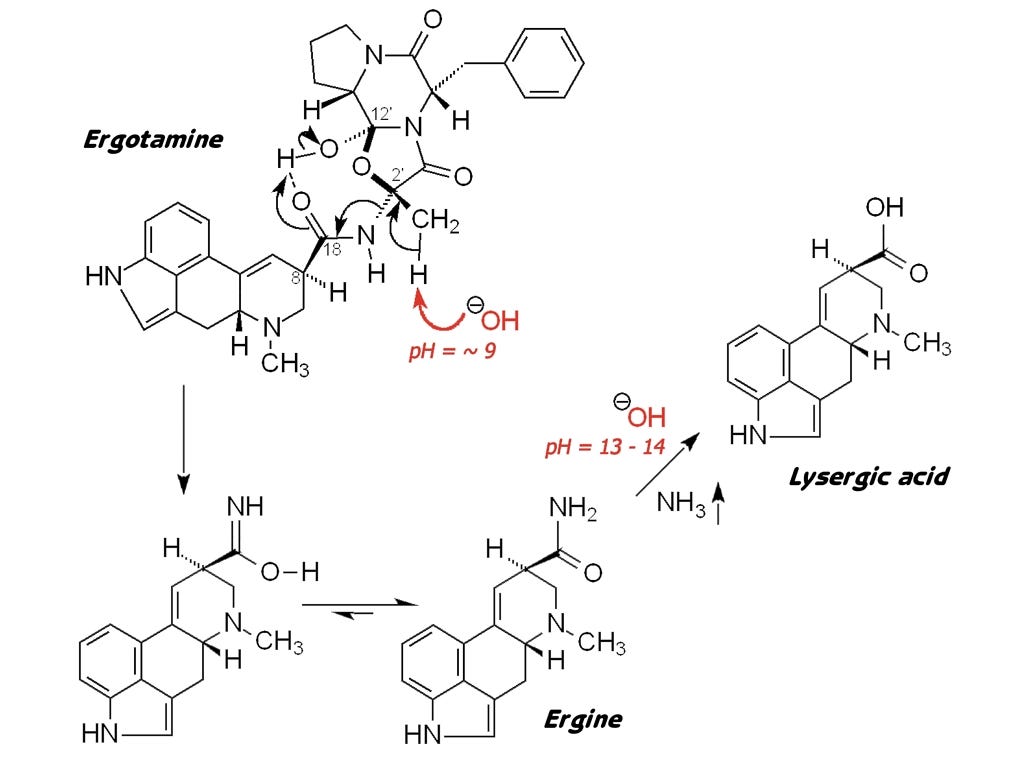
Here we see the reaction mechanism proposed by Dave Nichols and Dan Perrine for our paper in ELEUSIS. The first stage of this reaction, the partial hydrolysis of ergotamine and its chemical cousins to ergine, can be brought about by fairly mild basic conditions, such as a slurry of wood ash in water might provide. Only in much stronger basic conditions and higher temperatures does the hydrolysis proceed to the next stage to produce lysergic acid itself. The kykeon recipe we propose in the ELEUSIS paper suggests precisely this: finely ground ergot would be digested for a time in a mixture of wood ash and water and/or wine, probably at elevated temperature. Wood ash, of course, contains potassium carbonate, and stirred in water can easily produce a solution of at least pH 10 or even 12.
Now what makes this proposal doubly interesting is that ergine is the very same lysergic acid amide that had become famous as one of the shamanic drugs of ancient Central America. Ergine, the simplest lysergic acid amide, was the principle alkaloid of the seed of either of two morning-glory vines, called ololiuqui by the Aztecs of ancient Mexico. According to our proposals, therefore, the shamans of Mesoamerica and the priests of ancient Greece had discovered the very same psychedelic agent, albeit in two extremely different plants – one, a flowering vine, the other, a primitive parasitic fungus.
Our claim that ergine was the ingredient of interest in both ololiuqui and the kykeon would be a hypothesis that had some considerable advantages over the original suggestions in Albert Hofmann’s essay in The Road to Eleusis.
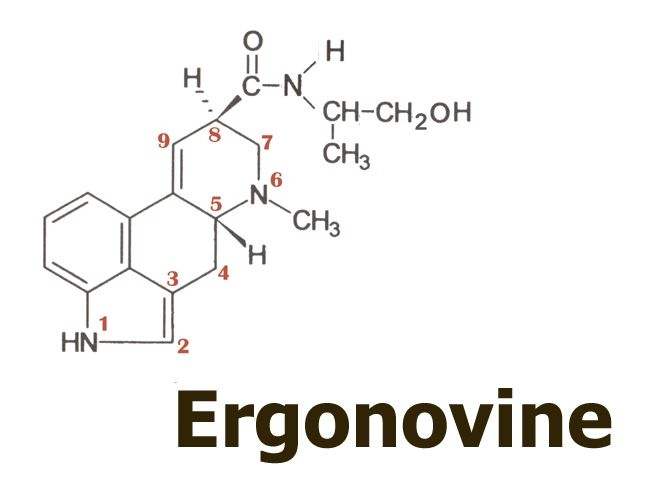
Dr. Hofmann had proposed that it might have been another simple lysergic acid amide, ergonovine
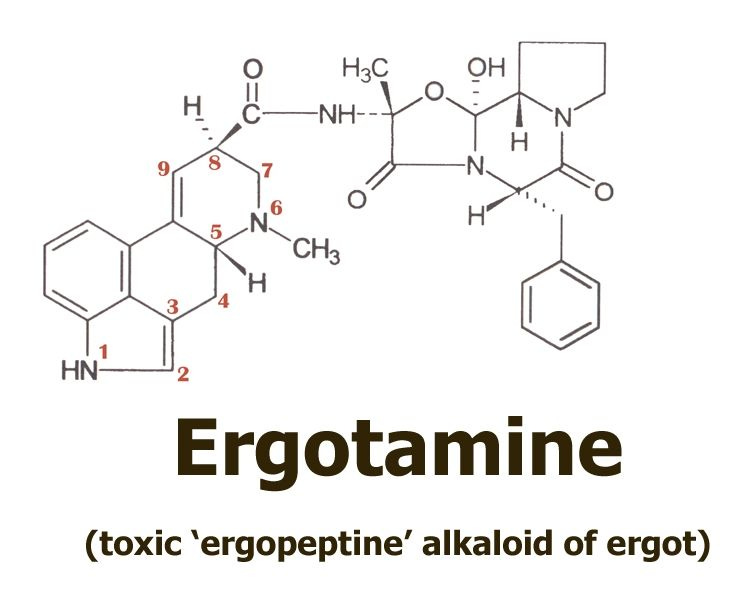
…that the Eleusis priests had learned how to obtain from ergot using a simple water extraction. According to this proposal, they might have thus eliminated the toxic ergopeptine alkaloids such as ergotamine,
…the presence of which was one of the important objections to the idea that ergot could have been the kykeon’s secret ingredient.
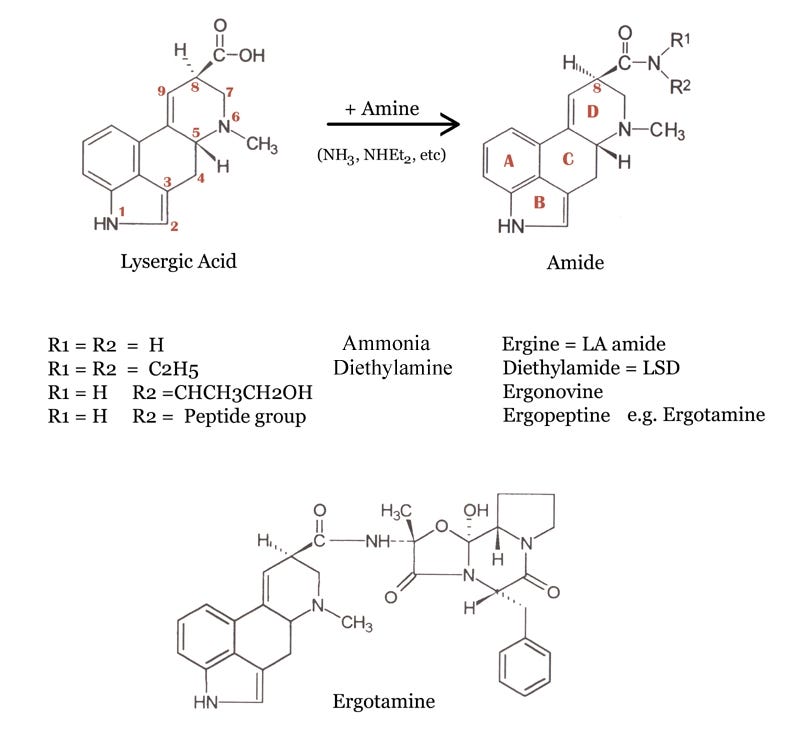
In these 3 diagrams you have just seen, you can easily observe how all these chemicals are derivatives of the basic structure at the left, the lysergic acid or ergolene base of all the ergot alkaloids. Here are the three chemicals once more, as an animation:
There were two remaining problems with Hofmann’s ergonovine proposal, however: firstly, self-tests by a few interested researchers revealed that ergonovine did not seem suitably or sufficiently psychoactive to fulfil the kykeon’s reputation. Secondly, ergonovine is a minor and quite variable component of ergot, making it doubtful that a psychoactive kykeon could have been prepared reliably for nearly two thousand successive years.
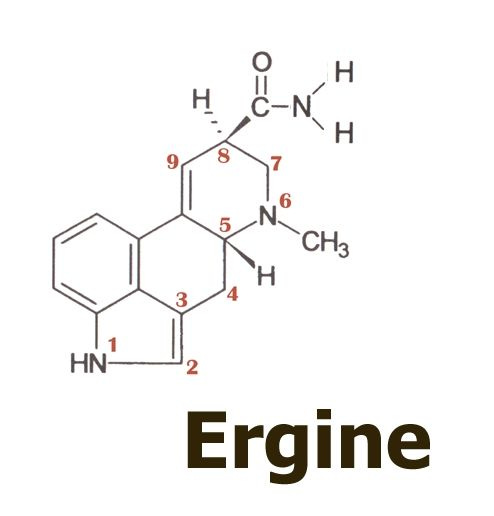
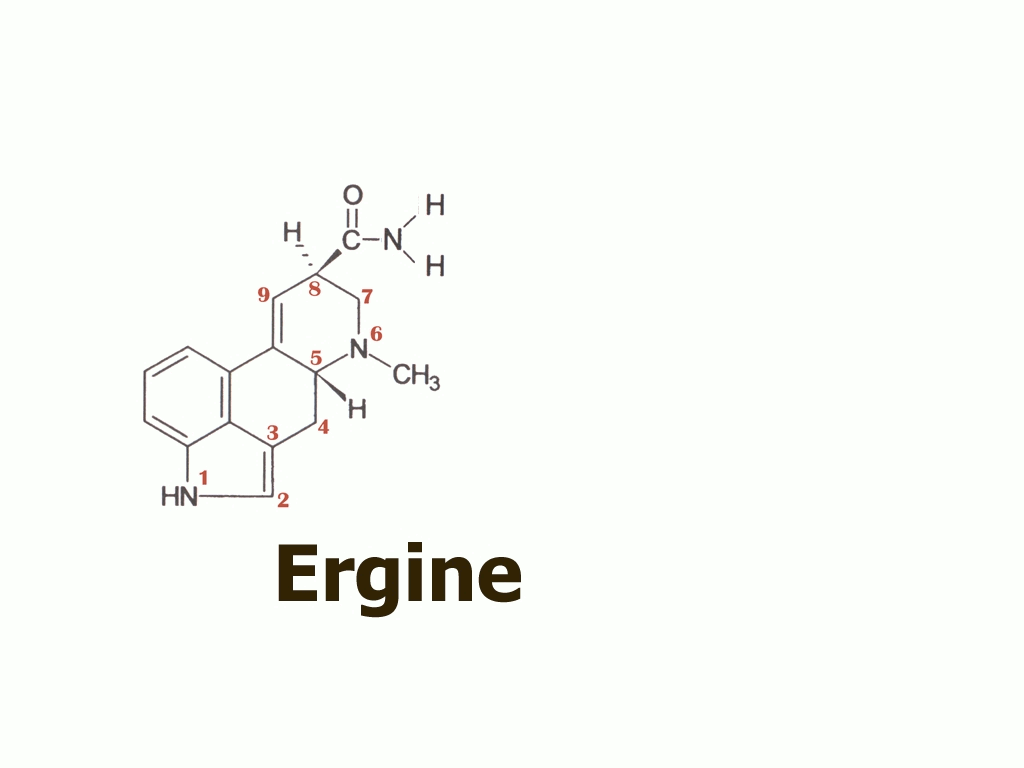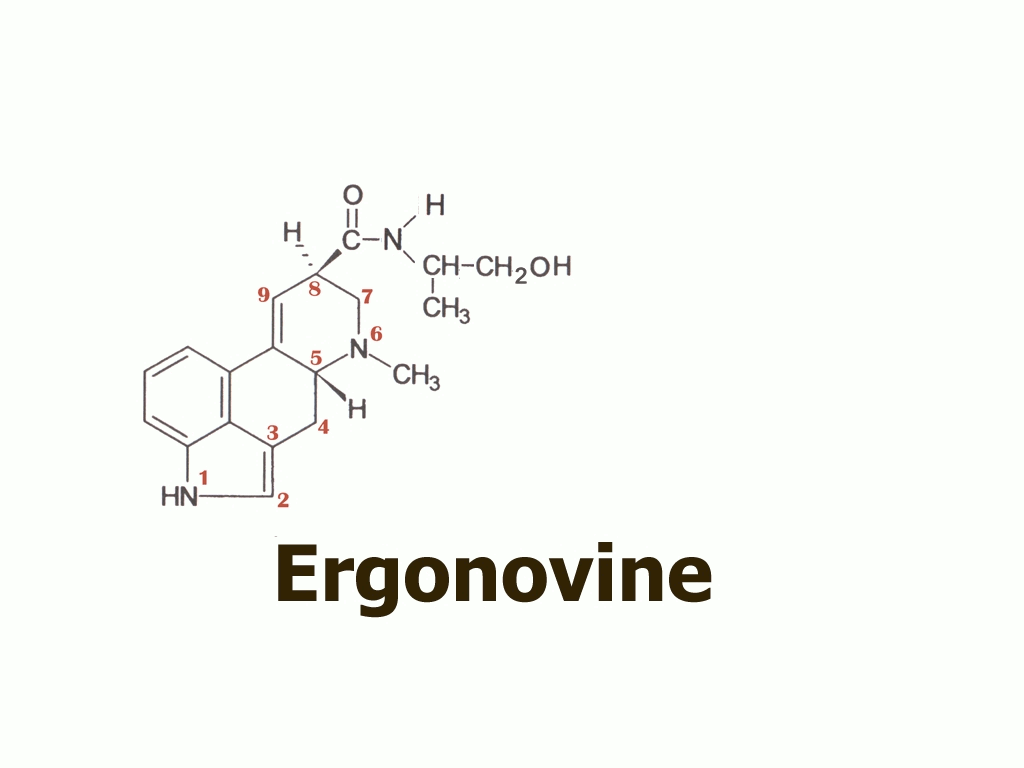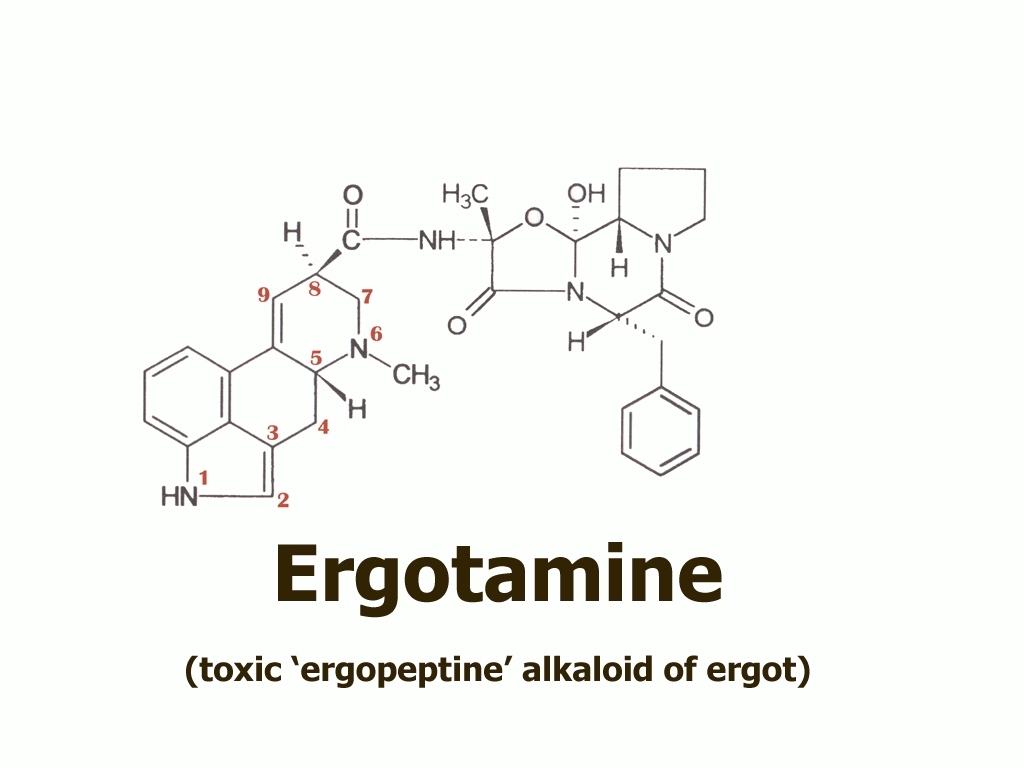
I would like now to take you all on a short guided tour of the lysergic acid molecule and its derivatives, particularly to show you some rather unusual characteristics of ergine, the simplest of the lysergic acid amides and the substance we now claim was the essential psychoactive component of the kykeon.
I have prepared a series of diagrams that will, I hope, make it possible for those who have only a vague understanding of organic chemistry to nevertheless get at least the drift of my arguments.
Here on the top left we see the base molecule of all the ergot alkaloids, lysergic acid. Note how the structure is numbered, and how the rings are labelled A through D. We will be most concerned with the D-ring of the molecule, and the way in which the hydrogen and amide group are attached to position 8.
The table shows the amides that result from the reaction of lysergic acid with various amines, to form amides. To synthesize ergine, we see that it is necessary only to react lysergic acid with the simplest amine, ammonia.
But to be more correct and specific, and as you will see in a moment, it is not just ergine but also its mirror image molecule, or epimer - isoergine - that enters the picture.
For there seems to remain a problem with the proposal that ergine might be the psychoactive compound of the kykeon, or even of ololiuqui. The problem is similar to that of ergonovine mentioned above – when some researchers have tested the alkaloid as a pure compound, it did not seem have the psychoactive qualities that make for an effective psychedelic. These tests were summarized in the second part of our paper in the ELEUSIS journal, “Mixing the kykeon“.
There have similarly been doubts that the ololiuqui of the Aztecs was a potent psychoactive, for self-tests using the seeds of the Ipomoea morning glory vine have also been inconclusive in some cases.
There have even been suggestions that the effect of ololiuqui was entirely due to suggestion. It does seem absurd, however, to suppose that the central American shamans would have employed a mere placebo when they had such a wide range of undeniably powerful psychoactives at hand, including Peyote and Psilocybe.
And, as Jonathan Ott has pointed out in a letter he wrote to us, “Ololiuhqui was far more prominent as an entheogen here in Mesoamerica than those mushrooms — the mushrooms are mentioned only here and there by a few competent chroniclers; yet almost an entire book was devoted to denouncing mainly the ololiuhqui idolatry. The annals of the Inquisition contain many times more autos de fe for ololiuqui than for mushrooms.”
So let’s take a closer look at ergine.
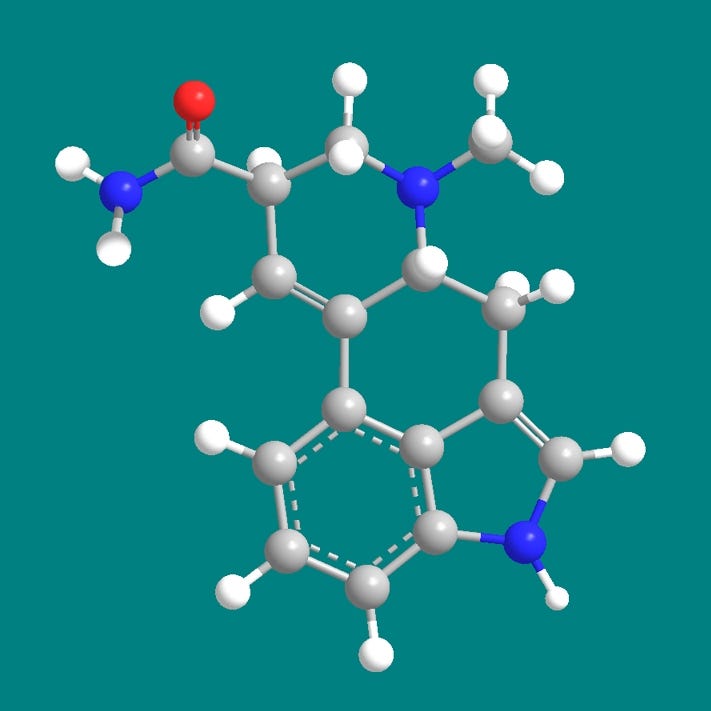
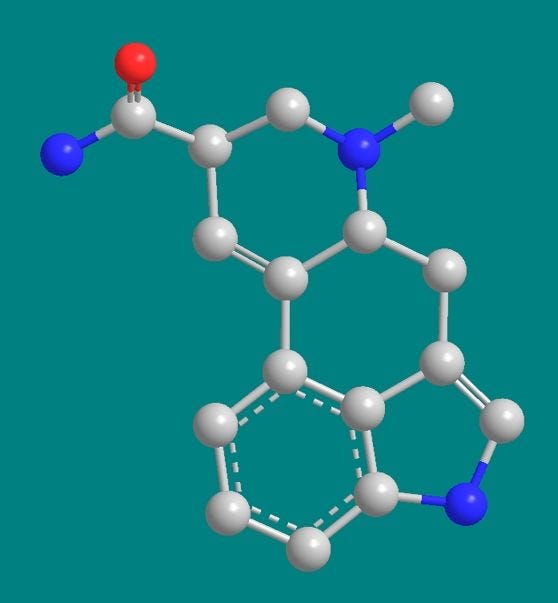
Here’s a model that should be easier to understand than the shorthand diagrams of the previous slide, especially if you’ve forgotten your organic chemistry lessons. Carbon atoms are in grey, and we can see how they form the backbone of the lysergic acid ring structure. Also joining in are nitrogen atoms in blue, hydrogen atoms in white, and a lonely oxygen atom in red. Now let’s simplify the model by ignoring the hydrogens, although let’s not forget they are there.
Now in this model it might first seem that the whole molecule is almost flat, but take a closer look at the bonds in the topmost or D ring. The attachment points of the bonds going to the blue nitrogen and the position-8 carbon atom imply that these atoms are behind the plane of the molecule. To see this better let’s first rotate the molecule clockwise…and then rotate the molecule again about its horizontal axis, with the top receding away from us.
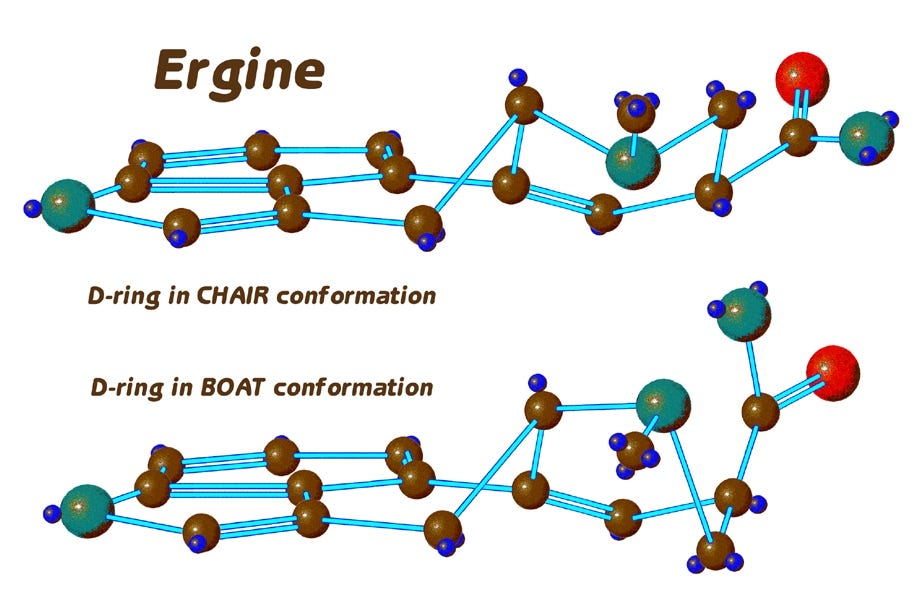
Now we can clearly see that the D-ring of ergine has a distinctly zig-zag shape whereas the rest of the molecule is basically flat, with all the carbons in one plane.
Let’s now go to some models I’ve drawn with somewhat exaggerated features that will better illustrate what is happening with the D-ring of ergine.
Here, the molecule is in the same position as in the last slide, but I’ve changed the colors and put back the hydrogens as small blue spheres adhering directly to their respective carbons. Ergine exhibits what might at first seem a bizarre behavior: not only does the D-ring have a decidedly zig-zag shape, it is constantly flipping back and forth to an alternate shape — or as chemists call it, a conformation, as we see here.
Let’s take a look at this conformation change in an animation, to more clearly understand what is happening. We see the D-ring vibrating back and forth between two different shapes.
Here are the two conformations side by side,
…and we designate them as the chair and boat conformation. The term “conformation” indicates that the forms differ only in their shape, not in the absolute positions of the atoms of the molecule.
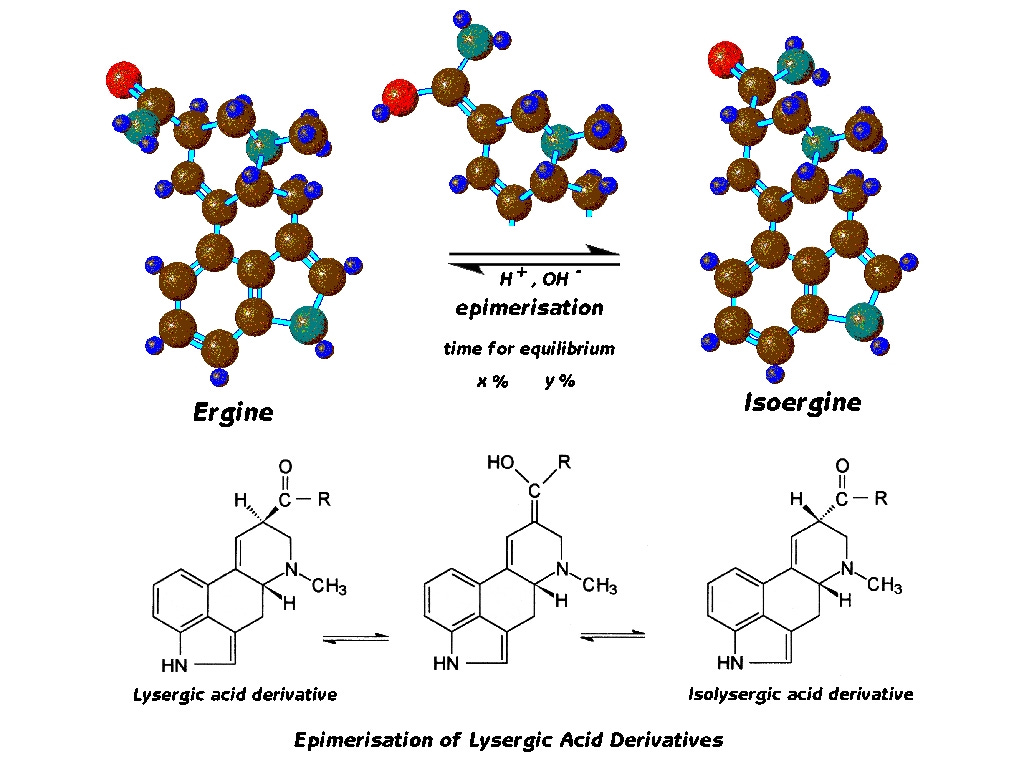
Now we must consider another change that ergine and all lysergic acid alkaloids undergo, and this also is a change in shape, but to arrive at the change two constituents of the molecule actually trade places.
The process is called epimerization, and proceeds through the formation of an intermediate compound we see here in the middle. The overall change brought about is merely the changing places of the hydrogen and the amide side-chain attached at the ring carbon atom in position 8. When this happens, we have the iso form of the alkaloid, and for ergine this is called isoergine.
The reaction is reversible, as we see by the double reaction arrows, and so after a certain time we can expect that the reaction will arrive at an equilibrium, and both ergine and isoergine will exist in the particular medium under consideration. The epimerization is thus typified in the case of each specific lysergic acid amide by an equilibrium concentration of the two epimers, and a time to arrive at this equilibrium. These factors are determined by the chemical and physical environment in which the molecule finds itself.
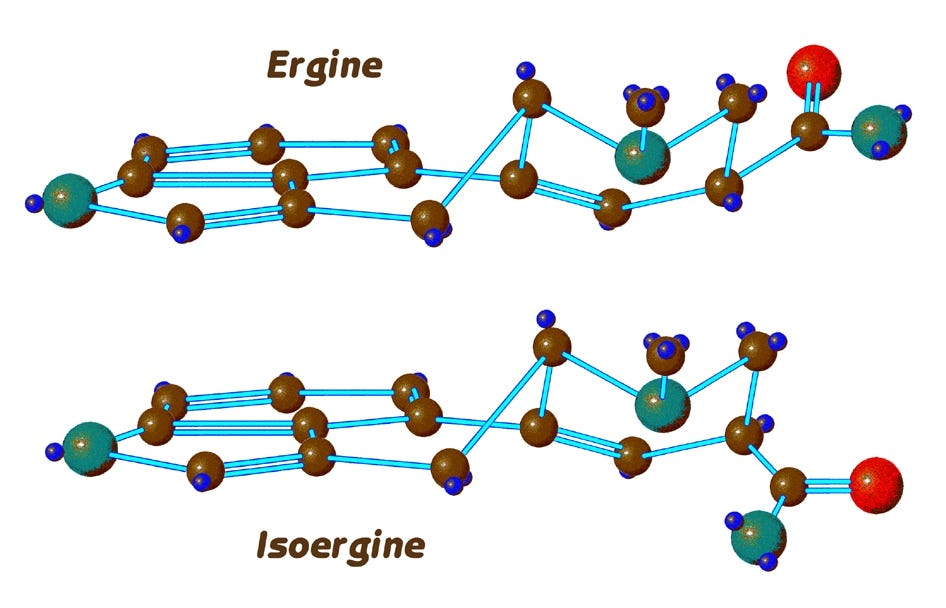
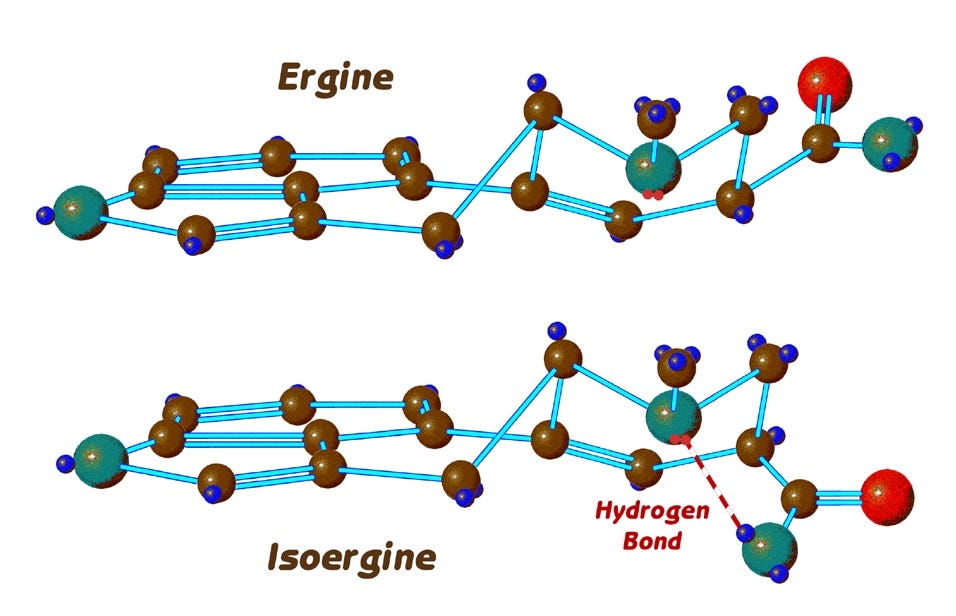
Here are the two epimers of ergine in profile, side by side as before. At the right, we now clearly see the reversed positions of the hydrogen and side chain attached at the 8-position ring carbon. Both ergine and isoergine here have their D-ring in the chair conformation we saw previously.
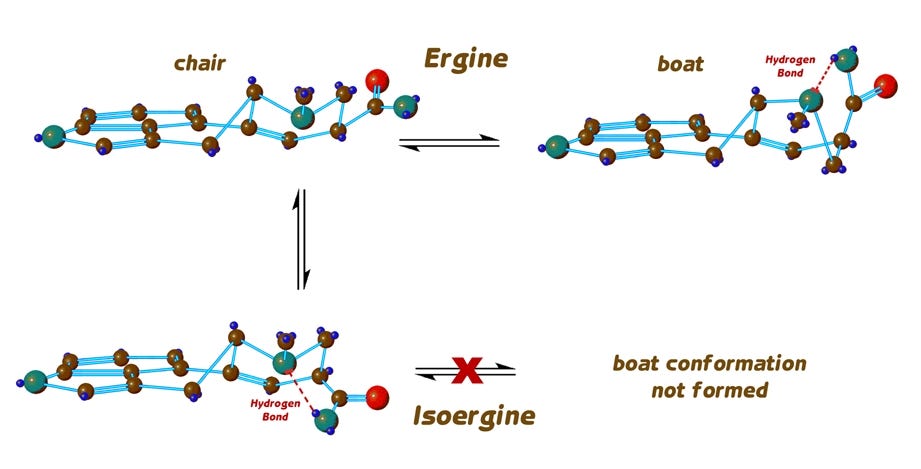
This brings us to a further complication that is specific to ergine but not LSD, and the question we must now ask is whether isoergine also exhibits flipping of its D-ring back and forth between the chair and boat forms that we see with ergine. In fact, isoergine does not readily change its conformation, for it is prevented from doing so by the formation of a hydrogen bond which locks it into the chair form.
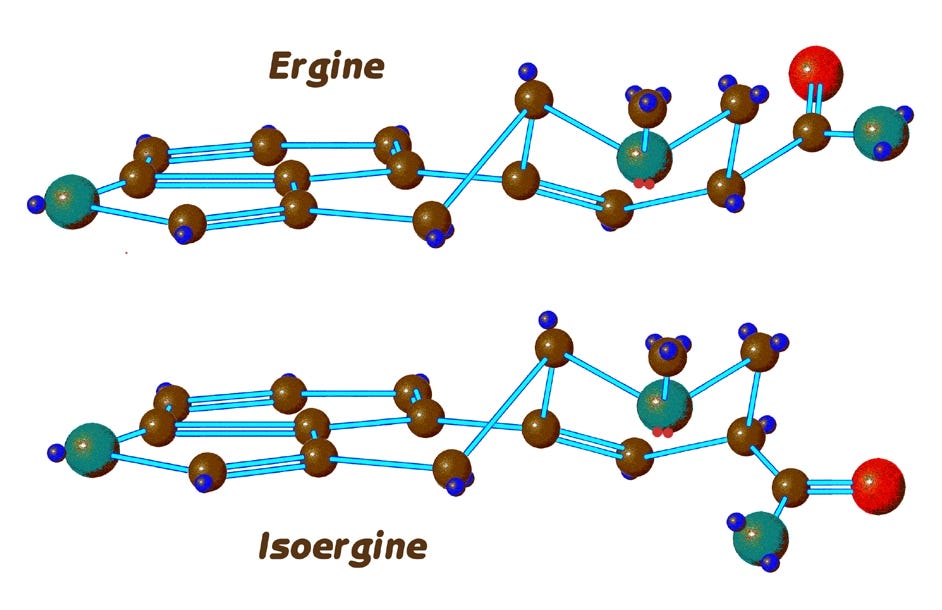
Here’s how that happens. I’m now going to add to the nitrogen atoms at position 6 of these diagrams what are called electron lone pairs. (The small red dots).
Nitrogen has rather more electrons than it uses in its normal three bonds, and these electrons cause one side of the atom to have a decidedly negative charge. In this case, when a hydrogen attached to another nitrogen is close enough to this negative charge, a hydrogen bond is formed.
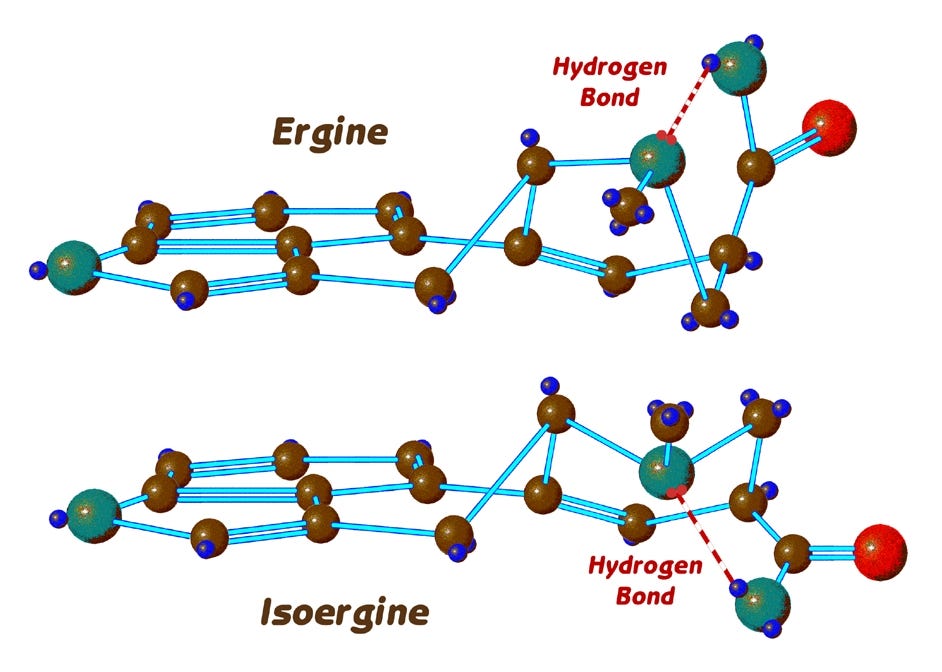
In the case of ergine, we can see from the top model that the corresponding hydrogen is too far away from the lone electron pair, and shielded from it by the poisiton-7 carbon. A hydrogen bond in ergine, CAN however form when it is in the boat conformation as we see here:
But since in general the boat form of lysergic acid compounds is less energetically favorable, the hydrogen bond in ergine does not prevent it from changing its conformation. All these results were confirmed in a paper in the journal TETRAHEDRON by Bernardi and Barbieri, using infrared spectra of the compounds.
So, we have the possibility that a molecule of ergine can be in one of three different states: It can be ergine in the chair or boat conformation, or it can be isoergine in the chair conformation. And in many chemical and physical situations, the three forms are constantly converting themselves from one to the others, and achieving a typical equilibrium distribution.
We therefore must consider that in an ergot preparation made according to our suggested method, and in morning glory seeds as prepared by Mesoamerican shamans, we are not dealing with pure ergine or pure isoergine. Both pure compounds have been tested and found wanting by some investigators, including Dr. Hofmann.
I would like to suggest therefore that the equilibrium mixture of ergine and isoergine may actually be the true psychoactive of the kykeon and ololiuqui as well. Since ergine spontaneously changes to isoergine when in solution, over a period of an hour or more, any process to partially hydrolyse the alkaloids of ergot such as our proposed recipe - or even the Aztec shamans’ procedure for extracting ololiuqui - should result in an equilibrium mixture of the three states represented here.
Taking either ergine or isoergine as a pure compound, however, may not result in the equilibrium mixture arriving at brain receptors. The equilibrium reaction takes some time to occur, perhaps an hour or more, and is brought about most effectively by basic conditions — and neither in the stomach nor in the blood do we find sufficiently basic conditions for the equilibrium to readily establish itself within a short time period.
Concerning the psychoactivity of ergine/isoergine mixtures, I have long had great confidence that extracts of morning glory seed, and by analogy a partial hydrolysis preparation made from ergot, could be quite powerfully psychoactive. In the late 1960s, when I started my research on these matters, I had gone to Mexico to experiment with morning glory seeds, and to begin with I extracted several kilos of seeds using a simple process, purifying an alcoholic extract between organic solvents in the alternating presence of aqueous solutions of ammonia and tartaric acid.
After a couple of days work, I obtained a nearly colorless syrup that exhibited the bright-blue fluorescence typical of active lysergic acid compounds.
A few of milligrams of this syrup, taken in a capsule, produced one of the most powerful psychedelic experiences I had known, and by then I had already taken high-dose LSD several times, as well as Owsley’s infamous STP (not recommended!), and the total alkaloid extract of peyote that I had prepared previously to the morning glory extraction.
Ever since, it has therefore been a mystery to me why ergine should be such a fickle psychedelic, failing with some trials yet succeeding in others. My explanation — perhaps not entirely satisfactory I admit — is that my extraction procedure allowed the equilibration of the original extracted ergine to the three ergine variants, and it was this mixture that was so effective. We do know, of course, that sometimes a mixture of two or more drugs can be more effective than any single component of the mixture alone.
A question kept popping up in my mind, however. Why shouldn’t ergine itself be reliably psychoactive? It is a close relative of LSD and its cousin, the dimethylamide, both of which are undeniably and strongly psychedelic. Perhaps this flipping of ring D between chair and boat conformations made ergine less available to their target brain receptors — or even prevented ergine from remaining at receptors known to be affected by LSD. There are other examples of drugs whose receptor affinity is limited by such conformation change, so it seemed a good bet that this might be the case with ergine as well.
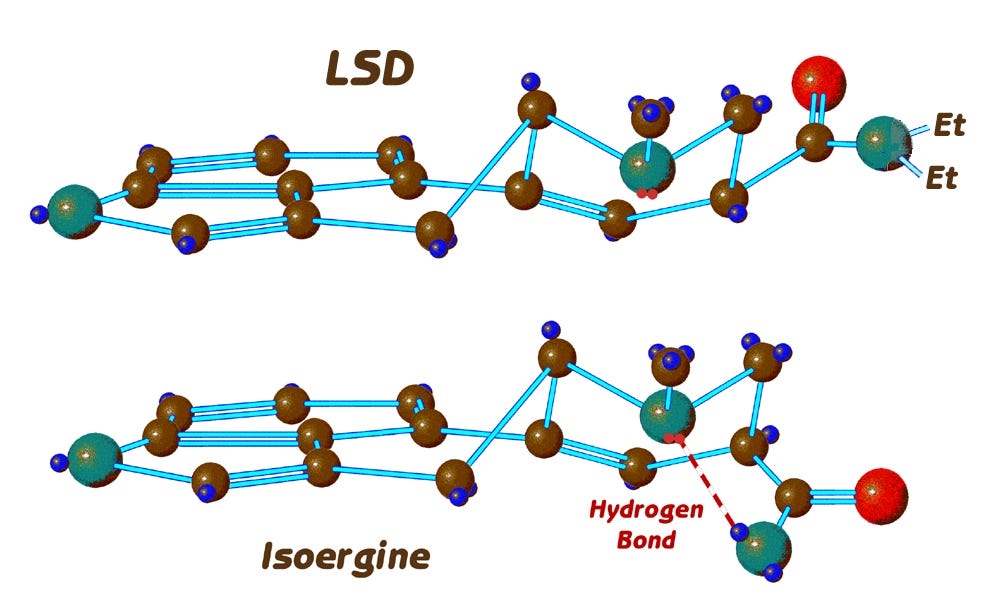
LSD, having ethyl groups and not hydrogens on the amide nitrogen, cannot form the hydrogen bond which stabilises the boat form of the D-ring as in ergine, or which stabilises isoergine to a unique conformation, so LSD remains exclusively in the chair form, and this might help to explain its extraordinary potency. And perhaps isoergine, like LSD, by virtue of its NOT changing conformation, actually was psychoactive, in spite of its being an iso derivative expected to be inactive.
Recently I enquired of Perplexity AI about the relative psychoactivity of ergine vs. isoergine, confirming that isoergine is a superior psychedelic agent.
An additional consideration about the psychoactivity of LSD compared to ergine is also significant. It is in fact the stabilization of isoergine in the boat conformation by the hydrogen bond that affects the equilibrium concentrations of the two epimers, ergine vs isoergine. But since there is no hydrogen bonding possible with LSD to stabilize its iso form, the equilibrium concentration of LSD vs. iso-LSD is far from 50/50. In fact, LSD in water solution or the body is 88% in the active epimer and only 12% iso-LSD. In addition, it takes a very long time, up to a week, for pure active LSD to epimerise to that equilibrium concentration, so that a dose of pure active LSD would arrive in the brain intact, with essentially NO conversion to its epimer iso-LSD.
Tetrahedron, 1965. Vol. 21. pp. 2539 to 2551